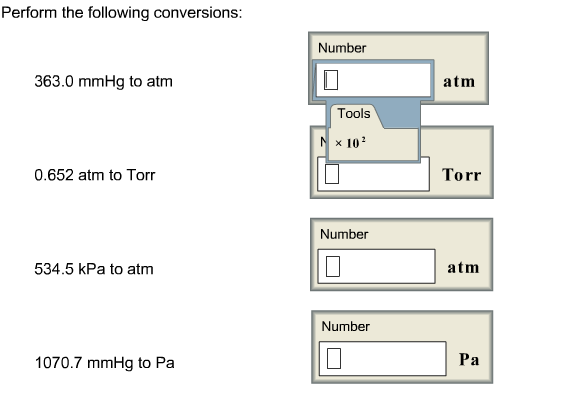
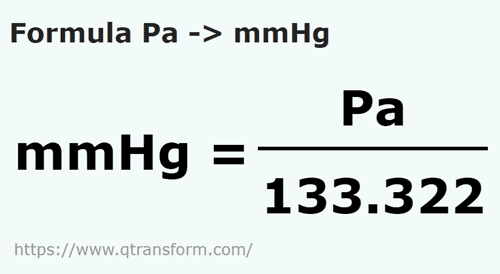pressure, conversion units into defferent units,atm,bar,torr,psi,Pascal,mmHg, numerical,and examples - YouTube

Gas Pressure Unit Conversions - torr to atm, psi to atm, atm to mm Hg, kpa to mm Hg, psi to torr - YouTube

Total pressure (1 mmHg 5 133.3 Pa) in the shunt for: (a) the optimum... | Download Scientific Diagram

Gases Copyright © The McGraw-Hill Companies, Inc. Permission required for reproduction or display. - ppt download

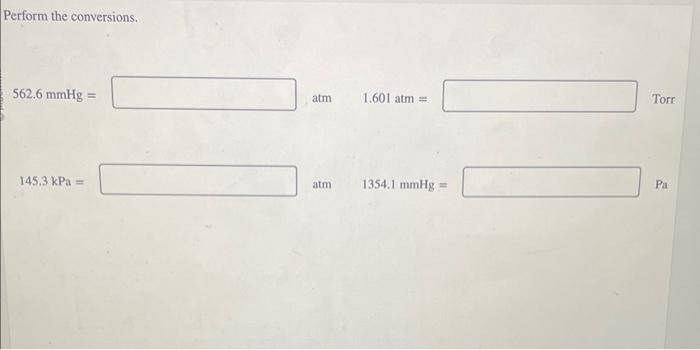
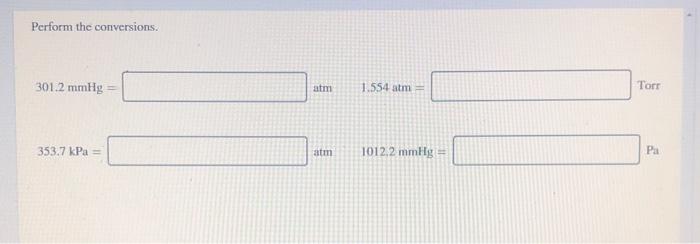
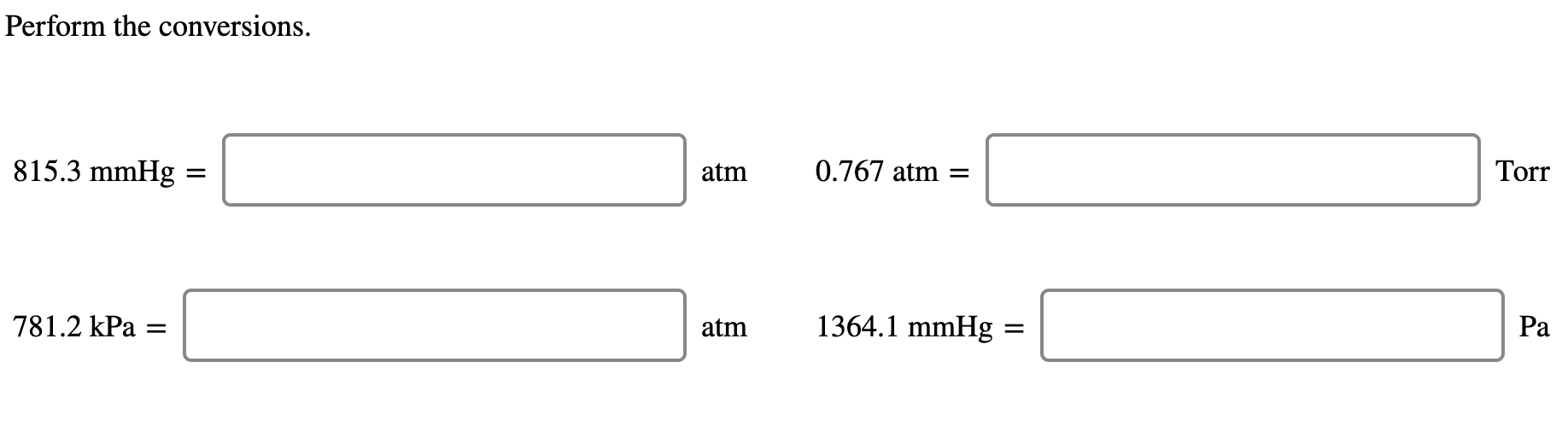
SOLVED: Exercise 2 a) Convert the following pressure to Pa, torr, mmHg, and atm: 25 psi 42 psi 75 psi b) Convert the following pressure to psi and atm 250 KPa, 22.7 Pa 230 mmHg, 385 torr

SOLVED: A student reads a barometer in the laboratory and finds the prevailing atmospheric pressure to be 709 mm Hg. Express this pressure in atmospheres, kilopascals, torrs, pounds per square inch, and

SOLVED: Convert 661,000 pascal, pa into torr 1 atm 760 torr = 760 mmHg 101.,325 pa 101.325 kpa 1.01325 barr 14.696 p.si 29.921 inHg (inHg and p.s.i. are approximations of the metric pressures and are subject to significant figures)